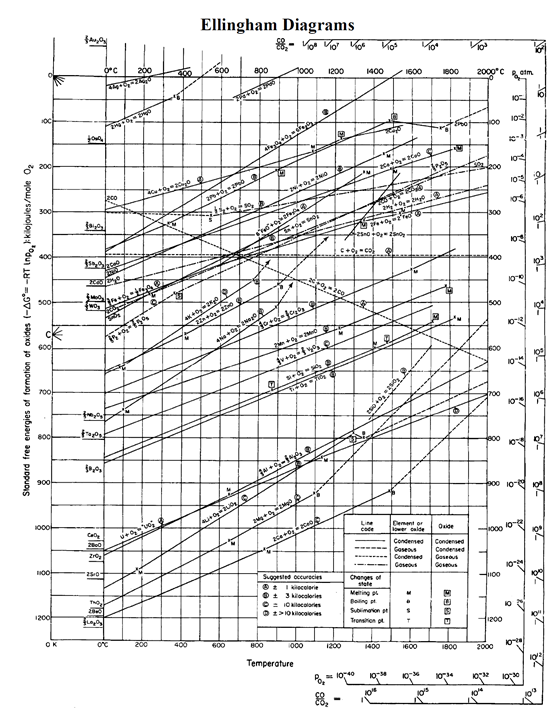
Thus the bronze age preceded the iron age the latter had to await the development of technology capable of producing higher temperatures, such as the blast furnace. The full working principle of an Ellingham diagram is explained in our thermodynamic training program ( Download the colorful InsPyro Ellingham diagram with H 2/H 2O axis here (download pdf).\) illustrates why the metals known to the ancients were mainly those such as copper and lead, which can be obtained by smelting at the relatively low temperatures that were obtainable by the methods available at the time in which a charcoal fire supplied both the heat and the carbon. H 2 use for reduction immediately impacts the atmosphere conditions and the reduction potential can not be maintained. Ellingham diagram representation of graphical (a) G vs T (c) G vs p 274. As long as carbon is present in the process, the CO/CO 2 ratio is kept low. These reactions generally involve the reaction of a gaseous phase. Originally the values were plotted for oxidation and sulphidation reactions for a series of metals, relevant to the extraction of metals from their ores (extraction metallurgy). When comparing the reduction potential of hydrogen to carbon, it is important to see that CO is a product from carbon. The Ellingham diagram plots 1 the standard free energy of a reaction as a function of temperature. If the ratio is larger Cu 2O will reduce to Cu metal, if the ratio is smaller Cu will oxidize to Cu 2O. Extend this to the H 2/H 2O scale (dotted line).Connect the H point on the left (orange circle), with the point 1200☌ on the 4Cu + O 2 -> 2 Cu 2O line (blue circle).In the Ellingham diagram, this can be read as follows (Cu example again): The higher the ratio, the more reducing the gas. The reduction potential will depend on the H 2/H 2O ratio in the atmosphere of the process. If the ratio is larger Cu 2O will reduce to Cu metal, if the ratio is smaller Cu will oxidize to Cu 2O.įor H 2 a similar reasoning can be made. Extend this to the CO/CO 2 scale (dotted line).Connect the C point on the left (orange circle), with the point 1200☌ on the 4Cu + O 2 -> 2 Cu 2O line (blue circle).Find the 4Cu + O 2 -> 2 Cu 2O line (orange line in the upper part of the diagram).Let us take copper as an example in a process at 1200☌: If you are interested to know which CO/CO 2 ratio leads to a reduction of your metal, you can use the Ellingham diagram. The higher the ratio (higher CO or lower CO 2) the more reducing the atmosphere is. In industry, the CO/CO 2 ratio is often used to indicate if a process is in a reducing or oxidizing state. The more negative the Gibbs energy is (= the lower the line in the diagram), the more stable the oxide becomes. 
The vertical axis gives the Gibbs energy of the oxidation reaction. Every line in the diagram represents the oxidation of a metal to a metal oxide. In this diagram, the stability of different metals and their oxides are compared. The main tool for studying oxidation/reduction is an Ellingham diagram. Although the principle is rather straightforward, it is important to understand that thermodynamic boundaries limit the usage of carbon/hydrogen. Hydrogen binds with oxygen (from metal oxide) and leaves the process as H 2O. When carbon is replaced with hydrogen the principle remains the same. The principle is rather simple, carbon binds with oxygen (from metal oxide) and leaves the process as CO or CO 2 gas. Practically all processes use a carbon source to control the reduction extent. Successful metal-making largely depends on the art of controlling the oxidation and reduction reactions. Not only to replace fossil-fueled energy, but also as a reductant in metallurgical processes. Download the colorful InsPyro Ellingham diagram with H 2/H 2O axis here (download pdf).ĭue to the need for decarbonization in the manufacturing industry, hydrogen comes into the picture as a potential green alternative.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
